Animal Health Council Report November 2025

TB
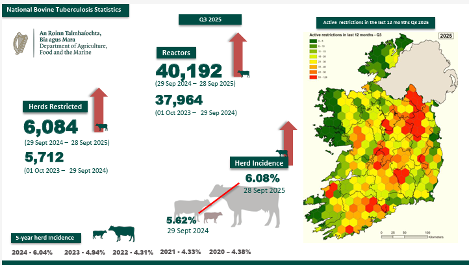
The TB action plan was launched by the Minister for Agriculture Martin Heydon on the 9th of September. The last engagement IFA had was a meeting held with the Minister on the 22nd of May. After this meeting a letter was sent to the Minister on the 3rd of June from the President outlining IFA’s position along with comments on the proposed measures. This was the last formal communication until the launch of the TB Action Plan. The TB Action plan lacks details on how measures will work and be implemented at farm level. Further engagement is needed with DAFM to iron out the details of each proposal. There is no date confirmed yet as to when these new measures will be in place.
The TB Action Plan



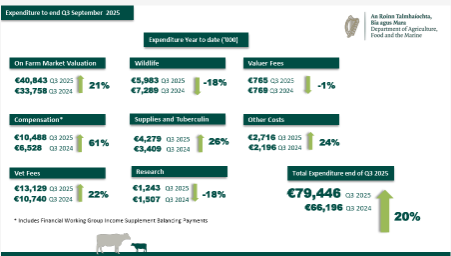
Budget 2026 has allocated €157m to the TB programme, an increase of €85m on the allocation for this year.
This year’s programme is estimated to cost €130m in total, requiring a significant supplementary allocation to Budget 2025’s €72m.
The breakdown of the expenditure can be seen in the tables above.
IFA Reaction
- Significant progress in proposals to address wildlife
- Some measures will be difficult on famers and must be quantified
- More detail needed from DAFM on some of the proposals
- Will not reduce TB levels if;
- Funding and resources are not provided to implement the programme effectively and consistently throughout the country
- All compensation schemes must be reviewed for fitness for purpose
- First issue for the Minister to resolve is the outdated arbitrary ceilings of €3,000 and €5,000 in the live valuation scheme
BVD
There is an increase in BVD rates year on year with some large breakdown on farms this spring. The reduction in disease prevalence and vaccination has led to increasingly naive herds. DAFM must ensure that there are increased levels of vaccination available for farms surrounding outbreaks. Irish farmers have worked to reduce this disease to near eradication, and this progress must be protected. In 2025 DAFM provided €3.75 million equating to €2.40 per calf for a max of 25 calves.
IBR
AHI proposed a program that will cost €40 million per year and have a 16-year timeline to achieve eradication, along with some movement controls on farms. The funding model remains the critical issue that must be addressed to advance a programme.
Johnes
Phase two of the Johnes programme ended in 2023. IFA is currently engaging with DAFM, ICOS and AHI to develop a funding model for the next phase of the programme.
IFA is seeking full financial and technical support for farmers with herds positive for Johnes. The BTM screening for Johnes has proven to be an effective screening tool for all dairy farms in the country. AHI has proposed a new scheme for farmers that have shown 2 BMT positive in the last three years. The proposed funding is €550 in year one, €550 in year two, €150 in year three and €150 in year 4. The current administrative delays in farmers being informed of a BTM positive test is hindering any progress in this scheme being launched with spring 2024 letters being sent recently and the majority of autumn 2024 and spring 2025 letters informing farmers of a positive test still outstanding.
Veterinary Medicines
The Minister for Agriculture has signed the Veterinary Medicinal Products Regulation
- It is to come into effect in December 1st, 2025, for Antiparasitic products
- Until then Antiparasitic products can continue to be purchased without prescription
- IFA engaged proactively with all stakeholders throughout the process and continue to raise concerns with the recently signed Regulation
- IFA made a detailed submission to the Minister and his officials identifying the key issues to be addressed in the Regulation which was broadly supported by all stakeholders.
- Antiparasitic products became Prescription only Medicines following the publication of the EU Veterinary Medicines Regulation in 2019 and subsequent HPRA review of these products.
- Prior to this Ireland were the only country in the EU where these products were not POM’s
- The SI provides for vets on behalf of licenced merchants and Co-ops to prescribe for these outlets on completion of a Proper Assessment Protocol
- The farmers own vet will continue to prescribe based on their knowledge of the farm, and the animals on it as is current practice.
- There is a missed opportunity to broaden the supply base for farmers sourcing vaccines that are categorised as POM(E) medicines.
- These products have always been available only through veterinary pharmacies and vets without the need for a prescription and will continue to be available through these outlets.
- The validity period for Prescriptions for non-antimicrobial products has been set at 6 months
NVPS
During meetings with DAFM on NVPS, IFA raised several issues. The need for an on-farm supply of emergency use medicines, an agreement previously secured by IFA. The lack of visibility of the prescription to farmers, the timeline that prescriptions must be uploaded to NVPS and the flexibility to part fill prescriptions. DAFM has again accepted these needs. DAFM will provide a full prescription on AIM and a more detailed text message with product name and quantity. The last official report received on the number of prescriptions that have been uploaded onto NVPS is around 25% of those wrote to that point. DAFM have delayed the introduction of Veterinary Medicinal Products Regulation for antiparasitic products until the 1st of December 2025
Bluetongue
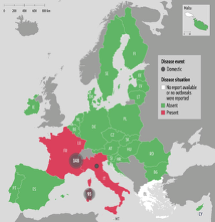
Bluetongue serotype 3 (BTV-3) was detected in the Netherlands in 2023 and has successfully overwintered in several countries in 2023 and 2024. A new serotype BTV-12 has been detected in The Netherlands in October 2024 with one case detected in the UK in February 2025. Genetically, it is unrelated to BTV-3 with its origins currently unknown. Other strains of Bluetongue stereotypes, BTV-4 and BTV-8, continue to circulate in other European countries with BTV-5 recently detected in Italy. A more virulent strain of BTV-8 emerged in France in August 2023 with cases in France and Spain in 2024 and 2025. This strain has more saver health implications with a high mortality rate amongst sheep flocks. There are approved vaccines available for both BTV4 and BTV-8 with different approaches being taken on the administration and cost burden in different countries.
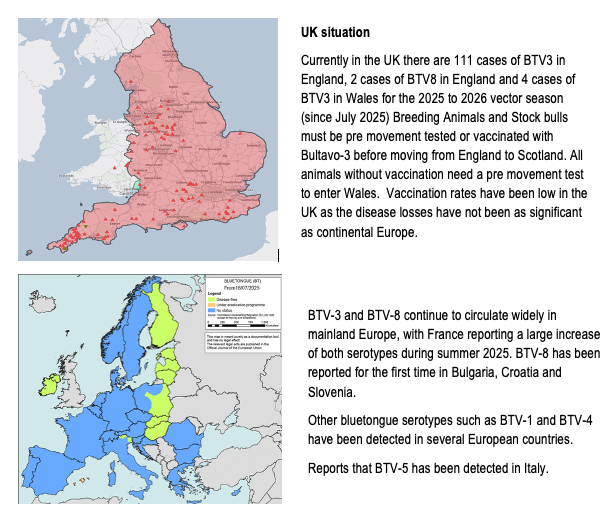
The Department would likely establish an eradication programme for Bluetongue virus. Certain control measures would be put in place in a 150km zone around the outbreak, or the whole country, depending on location.
Measures may include but are not limited to:
- Surveillance of infection of Bluetongue virus
- Movement restrictions of the target animal population within and from these zones
- Vaccination of the target animal population (if available)
- Humane destruction of infected/suspected animals, if necessary to prevent spread of the disease
- The movement of cattle or sheep would be controlled within the 150km surveillance zone
- Exports of live animals would be subject to restrictions outside the EU with derogations in place for within EU trade.
- Controls would be kept in place until there is no further risk of spread but may be relaxed during the period when the insect vectors are not active (the vector free period)
There is currently no licenced vaccine for BTV-3 but vaccination under emergency use authorisation is being carried out in the UK and EU. IFA ask that The Department immediately move to approve vaccines for use in this country and put in place a stock of these vaccines to have available for use should the need arise.
Lumpy Skin Disease
In June 2025, Italy reported a first outbreak of lumpy skin disease in a large cattle farm in Sardinia, followed by a second case in the Lombardy region. All affected animals were slaughtered. Also in June 2025, France reported the first outbreak of lumpy skin disease in a dairy cattle establishment in the Auvergne-Rhône-Alpes region, after observing the typical clinical signs in several animals. Spain has reported its first cases on 6th of October in the Catalonia region. Emergency protective vaccination in both countries has seen over 700,000 vaccines. administrated. Transport through these regions is facilitated for live exports.
New ER 1 Form (Herd number application forms)
IFA have had several meetings with DAFM on the new requirements for herd number applications. These changes were rolled out by DAFM in December 2024 without any consultation or discussion with IFA. IFA are seeking changes to the new application form in particular around the area of new applicants for herd numbers.




