Animal Health Reports
Animal Health Council Report June 2021

TB
- The Finance Working Group continue to meet regularly to develop the future funding model, to review the live valuation scheme and to review the current supplementary payments.
- The Implementation group was created to review DAFM programme proposals and implementation of the EU Animal Health Law requirements. The Implementation group had a meeting on May 11th which the chair decided to stop early due to a difference of opinion in relation to the terms of reference for the group. The basis of the decision to end the meeting early is inconsistent with the terms of reference for the group and so IFA have sought a meeting to address these issues and advance the work of the TB Forum Implementation group consistent with the agreed terms of reference. The next meeting of the Implementation Group is scheduled for the 8th of June.
- The latest meeting of the bovine TB Stakeholders forum was on Wednesday 26th May and included an update on the current TB situation, report from working groups, communications update, strategic review of the TB Programme and a review of the inconclusive policy.
- IFA have sought a response from the Data Protection commissioner, through the Association’s solicitor, to our complaint in relation to the Herd Categorisation letters issued by DAM in August 2020. The most recent communication (received 10thFebruary) from the Data Protection Commission states that the matter is under review and that contact has been made with DAFM.
- IFA continue to set out the key areas that must be addressed in the TB Strategy.
- These include:
- The revoking of the new herd categorisation coding system which came into effect with the TB herd Risk letters.
- The proposals for pre-movement testing
- The need for an enhanced Wildlife Control Programme
- Better investigation and resolution of TB outbreaks
- Fair Financial supports for farmers impacted by TB controls.
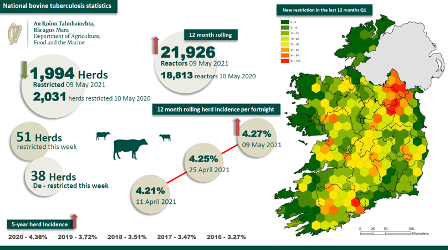
Latest bovine TB Statistics
BVD
- IFA support the objective to reach BVD Free Status by 2023, recognising the benefits and the investments of farmers to-date in the programme.
- IFA have identified and raised a number of issues with the new programme including; movement under permit, communication, tone and content of restriction letters and appropriateness of actions for restricted herds. Commitments to improve on these areas have been secured for the 2022 programme
- The next steps for the BVD Implementation group are to review the 2021 programme and to identify effective case detection high level surveillance after ‘Free Status’ is achieved.
Latest BVD Weekly Figures

Veterinary Medicines
- The new EU Veterinary Medicine Regulations were approved by the Commission in January 2019 and come into effect in 2022. IFA have identified categorisation, prescribing process and access to antibiotics as key areas of concern since 2015. IFA raised these issues with Ministers of Agriculture, MEP’s and senior DAFM personnel. DAFM refused to pursue a resolution to the issues identified by IFA.
- Key issues with the regulations include: Anthelmintics becoming POM in 2022, prophylactic use of antibiotics will be banned (Eg. Dry cow tubes), it will only be possible to prescribe exact quantities of antibiotics necessary to treat specific conditions, a prescription for antibiotics will only be valid for 5 days and the issue of who can prescribe.
- IFA position remains that SQP’s must be facilitated in prescribing and supplying anthelmintics. This position is supported by licensed merchants and pharmacies.
- All other countries in the EU categorised Anthelmintics as POM in 2007. Ireland availed of a derogation to exclude anthelmintics from POM category at the time. In the UK these products are classified as POM but have facilitated SQP prescribing. In the new regulation, NI can continue to avail of SQP prescribing. This will create a two-tier supply system will come into effect on the island. This creates an incentive for illegal trade across the border.
- The VCI prescribing guidance requires vets to have visited the farm inside the previous 30 days to prescribe antibiotics and 90 days to prescribe all other medicines and VCI also refuse to recognise laboratory results as adequate information to prescribe.
- The IFA position remains, laboratory test results must be accepted as adequate knowledge to prescribe similar to the current Schedule 8 operated by Co-ops in mastitis control programmes.
- Farmers must have access to important medicines to protect the health and welfare of our animals, limiting access to antibiotics as set out in the EU Regulation will have serious animal health and welfare implications.
- The potential for electronic prescriptions has advantages but IFA have raised data concerns with DAFM.
- The access electronic prescriptions provide for DAFM and others to individuals to farmers medicines usage is not acceptable, IFA have raised this with DAFM and sought a further meeting to address the issue
- IFA are actively pursuing a reduction in VAT rate on vaccines.
- IFA are currently involved in developing mechanism for farmers to dispose of unused antibiotics and other veterinary medicines.
Johnes
- Infection rates remain very low.
- The focus for 2021 is to encourage participation in the programme as participation remains very low.
- Anyone contacted by DAFM in relation to a positive Bulk Milk Test should not panic and should seek information from AHI and their vet and they should be encouraged to join the programme.




